Ionic Liquids (ILs) have shown to be a promising alternative to the state-of-the-art electrolytes for Li-metal and Li-ion batteries (LIBs). In an attempt to overcome the strong interactions between the IL anion and the Li+ cation and improve battery performance, we studied several “asymmetric” ILs by grafting two widely used anions into one molecule.
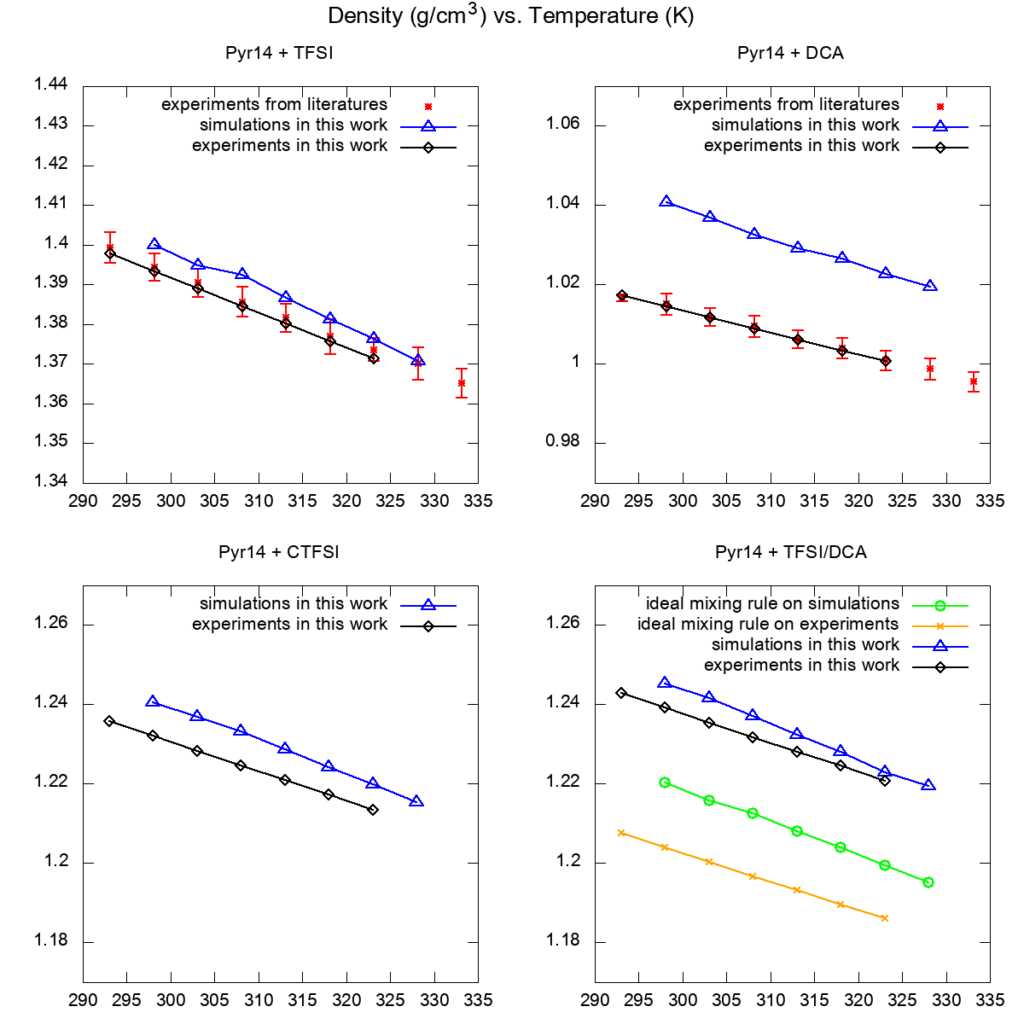
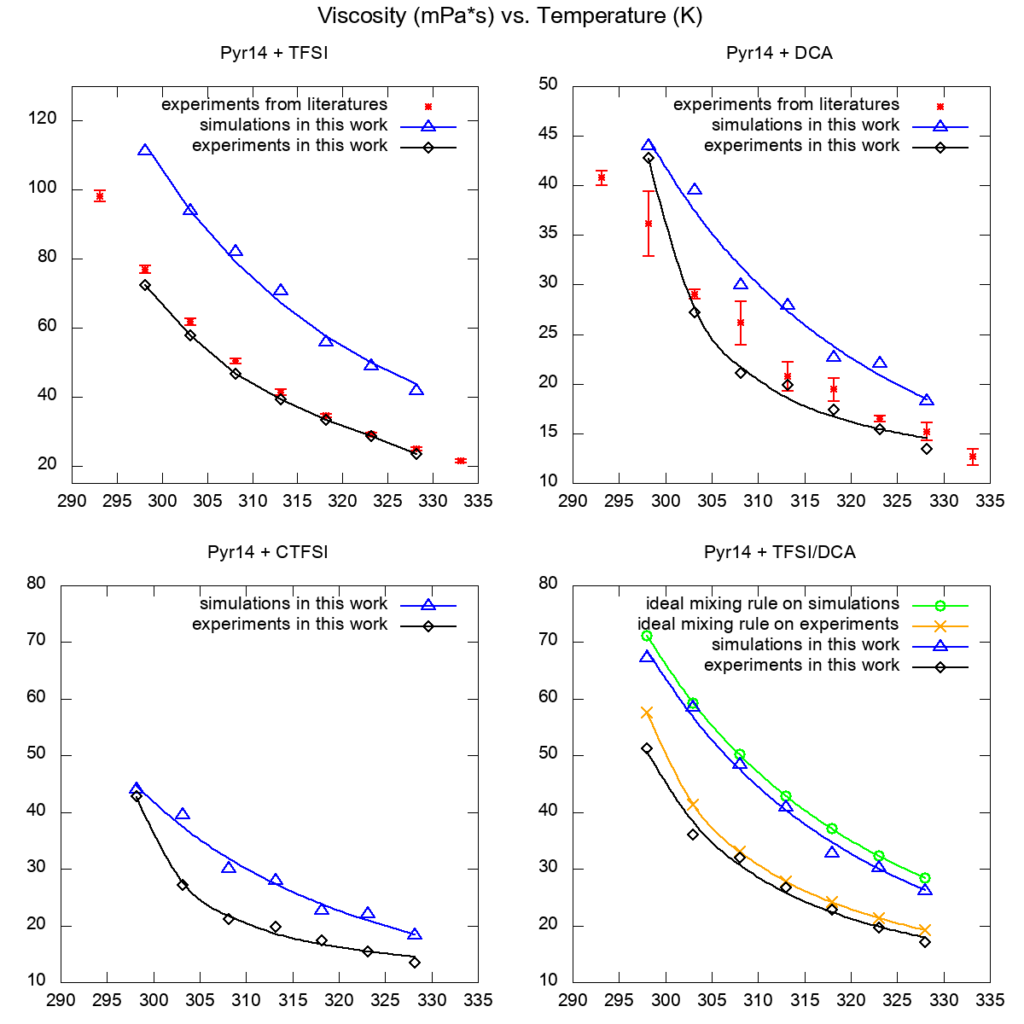
By comparing with experimental results in this work and in the literature, we found that our MD model agreed well in terms of densities and viscosities under different temperatures.
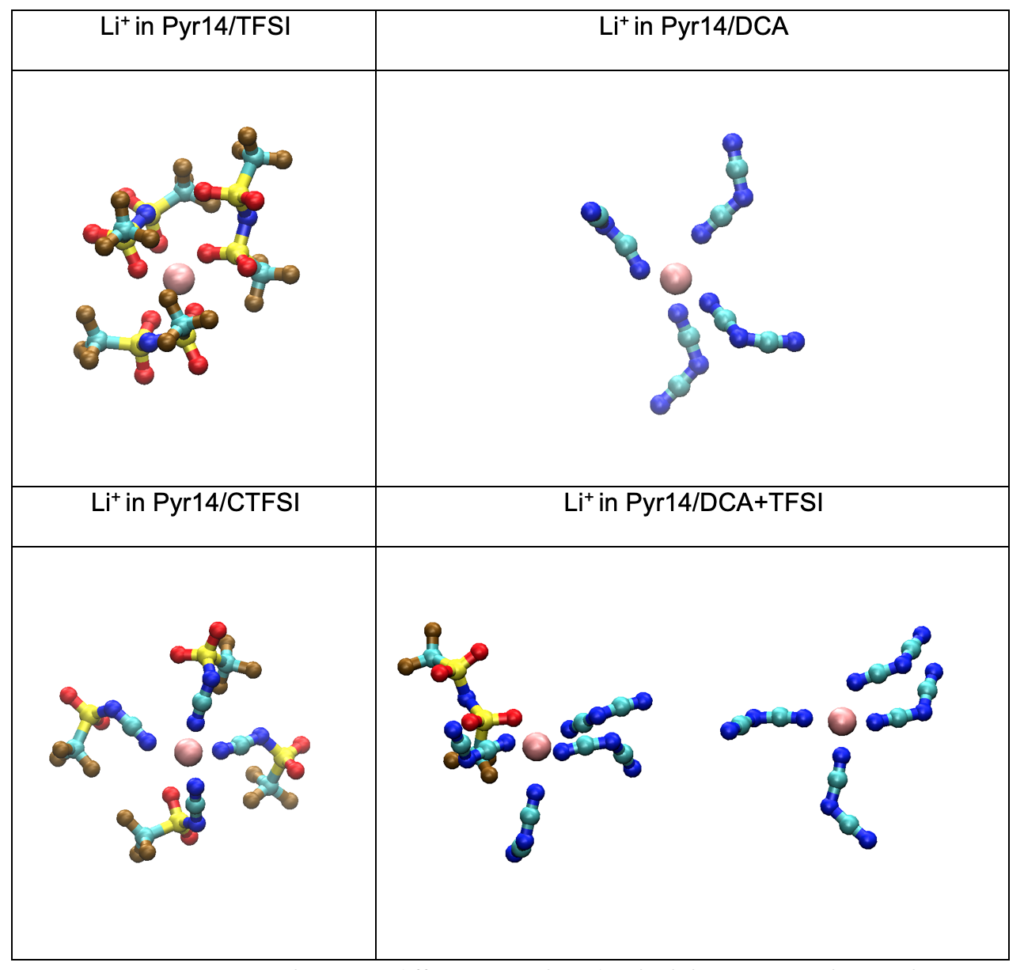
By using radial distribution functions (RDF) and coordination numbers (CN) of Lithium ions in different ionic liquids, we were able to provide optimal anion-to-cation cluster structures at molecular levels.
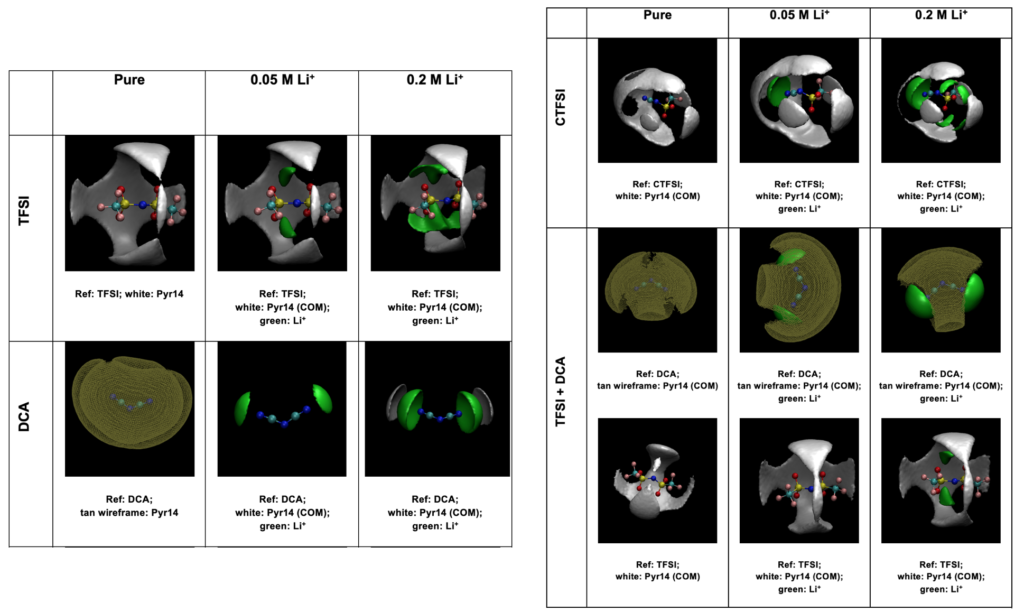
The spatial distribution was used to extract and show the information of how lithium or other cations are distributed around different anions. These are molecular events that are hardly captured by experiments, however, they are essential to elucidate ILs’ properties and provide guidance on designing new ILs.