Research Focus Areas
Custom Synthesis of Metal/Porous Nanoparticles
Nanotoxicology
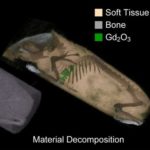
Biomedical Image Contrast Agents
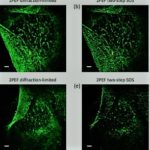
Precision Nanoprobes
Molecular Nanotechnology
Custom Synthesis of Metal/Porous Nanoparticles
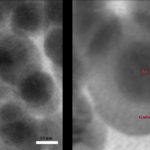
Standardized synthesis of Gold nanoparticles (nanoclusters, 5 nm, 10 nm, 16 nm) Silver nanoparticles (nanoclusters, 10 nm, 40 nm, 95 nm) Iron oxide nanoparticles (10 nm) Gadolinium oxide (5 nm, 10 nm) Hafnium oxide (5 nm, 10 nm) Microporous silica particles (15 nm, 50 nm, 100 nm, 400 nm) Other nanoparticles currently being optimized include …
Nanotoxicology
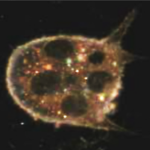
Nanotoxicity assessment is an integral part of designing biomedical probes. In vitro toxicity of nanoparticles is assessed using murine (L929) and human cell lines (HUVEC, RAW246.7). In vivo toxicity was assessed in zebrafish models, and in vivo biodistribution/blood pool kinetics was assessed in athymic and euthymic mouse models. Representative Publications …
Biomedical Image Contrast Agents
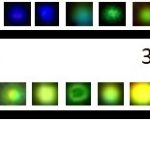
Scaled up the synthesis of nanoparticle contrast agents. I specialize in the synthesis of long emission lifetime, fluorescent and photoluminescent nanoparticles. This is achieved by modifying Au cores with fluorophore embedded shells or Au nanoclusters with tunable photoluminescence. Fluorescent silica particles with sizes ranging from 15 nm to 400 nm are also available. Such probes …
Precision Nanoprobes
Antibody-drug conjugates are emerging as a frontrunner in precision targeted therapeutics. However, this capability is still beyond the ability of most academic laboratories. The primary reason for this is the need to identify a precise binding site on the constant region of the antibody (Fc) for the drug molecule to be conjugated, while not affecting …
Molecular Nanotechnology
This is the current focus of my research. Utilizing anisotropic and hybrid nanoparticle architectures to introduce directionality in motion, precisely orienting targeting molecules and directed exertion of force by nanosystems. Anisotropic nanoparticles can be Janus hybrids or non-spherical nanoparticles and include rods, plates, prisms, and core-shells. This will lay the basis for nanomotors and nanomachines …
Military Health Sciences Research Symposium 2024. (Aug 26-29, 2024, Kissimmee FL)
Dr. Nallathamby presented on his abiotic biomimicking antibacterial Nanoparticles, and how they were successfully tested against A. baumannii BA1605 S.aureus USA300, P.aeruginosa FRD1, K. pneumoniae, C.striatum, E. faecalis, and S. pyogenes. We saw little to no evolution of resistance at doses above the MIC. Additionally, we observed what looks like inflammation-free wound healing in mouse and rat models. The wound size stabilizes as soon as the formulation is …
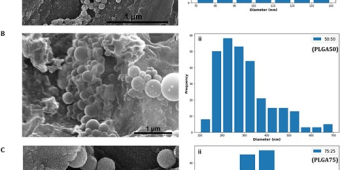
Sustained and Controlled Antibiotics Release for Prolonged Antibacterial Action Against MRSA, and Pseudomonas aeruginosa
Congratulations to REUs Argerie Guevara (NURF’21), and Kevin Armknecht (NURF’23) and Carlie Kudary (#BIPH) on the prolonged antibacterial action formulation of standard antibiotics to tackle MRSA and P.aeruginosa. More to come this year. #AMR #GOIRISH https://doi.org/10.1093/milmed/usae079



Recent Comments