According to Azra Raza, a Professor of Medicine at Columbia University in New York, high-grade brain cancer called glioblastoma is “one of the most aggressive, ruthless killers known to mankind”. Indeed, despite recent advances in cancer therapies, glioblastoma remains incurable with a median survival of 15 months which has not improved substantially in the last 20 years. This poor prognosis is, in part, due to the highly immunosuppressive microenvironment that allows tumors to evade anti-tumor immune response and promotes resistance to immunotherapy – a kind of therapy that uses your body’s own immune system to find and eliminate tumor cells.
Tumor immunity exists in a delicate balance between tumor-promoting and tumor-killing immune responses, and the ability to tip the balance towards the latter is often what makes an immunotherapy successful. Mechanical forces arising from tissues, including their extracellular matrix (ECM) components such as collagen and hyaluronic acid (HA) have been recently recognized as important regulators of immune cell function. Therefore, changes in ECM composition and mechanotransduction pathways that sense ECM stiffness offer novel therapeutic opportunities for glioblastoma patients. However, data from a recent phase I clinical trial targeting an important mechanosensing receptor CD44 using antibody therapy were rather disappointing with no evidence of clinical efficacy. The reason for failure may lie in the fact that CD44 may play antagonistic roles in regulating anti-tumor immune responses.
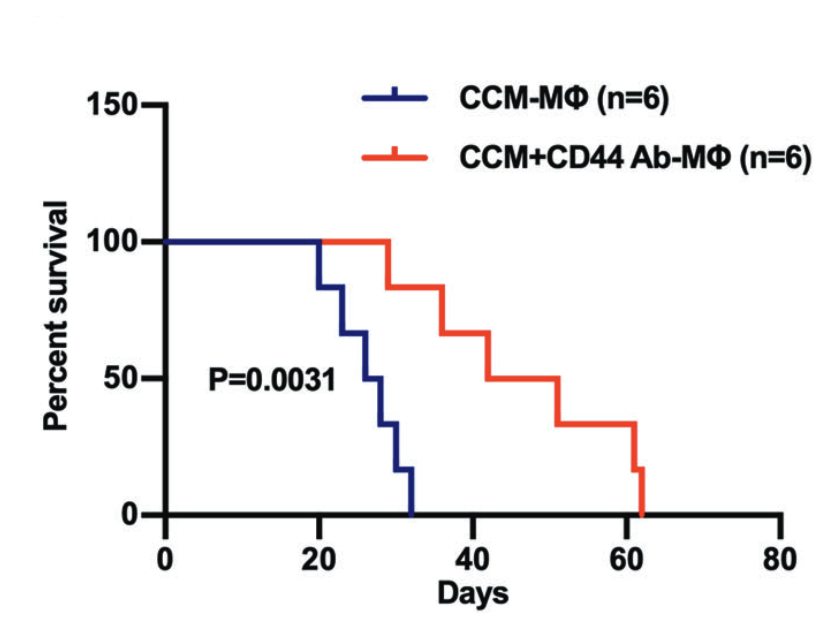
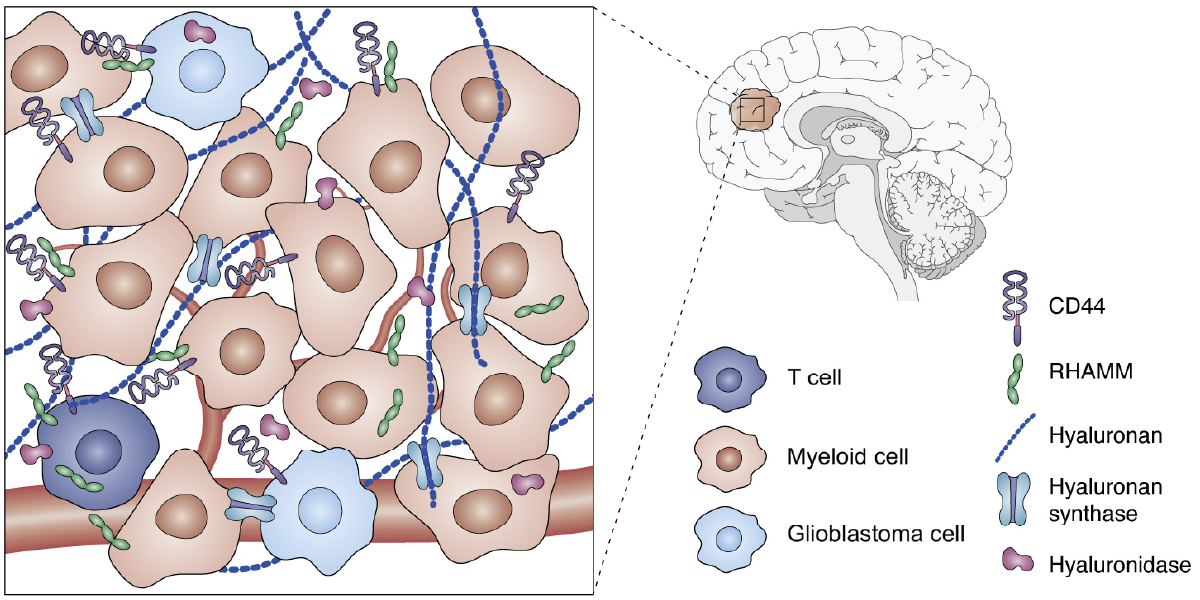
Myeloid immune cells are arguably the most important players in the glioblastoma microenvironment as they constitute the largest cellular fraction in most tumors. A recent study published in Nature found that HA produced by glioblastoma cells can signal through CD44 and reprogram the behavior of myeloid cells to promote tumor growth. The researchers showed that by blocking HA binding to CD44 on myeloid cells, they could promote the formation of tumor-fighting myeloid cells and increase the survival of tumor-bearing mice. A different study published later this year showed that CD44 expressed on myeloid cells also promotes invasion of glioblastoma cells into the surrounding brain. Because of its tumor-promoting role, CD44 may seem like a promising target for glioblastoma therapy. However, its roles in glioblastoma may be more tangled than it sounds at first.
T cells are immune system’s assassins that hunt down and destroy cancer cells. In T cells that infiltrate the tumor, CD44 receptors are commonly found on protrusions at the rear end of the cell that stabilize and move the cell through densely packed tissues. One study has found that CD44 was crucial for T cell navigation inside lymphoma tumors, and blocking or genetic removal of CD44 compromised T cell ability to infiltrate the tumor. Additionally, according to another study, CD44 plays an important role in interaction of T cells with another type of immune cells called dendritic cells that educate T cells to recognize tumor cells as foreign and dangerous. If CD44 is therapeutically inhibited like in the clinical trial of anti-CD44 antibody, this important line of communication between immune cells will be severed, and T cells will not be able to tell a friend from a foe.
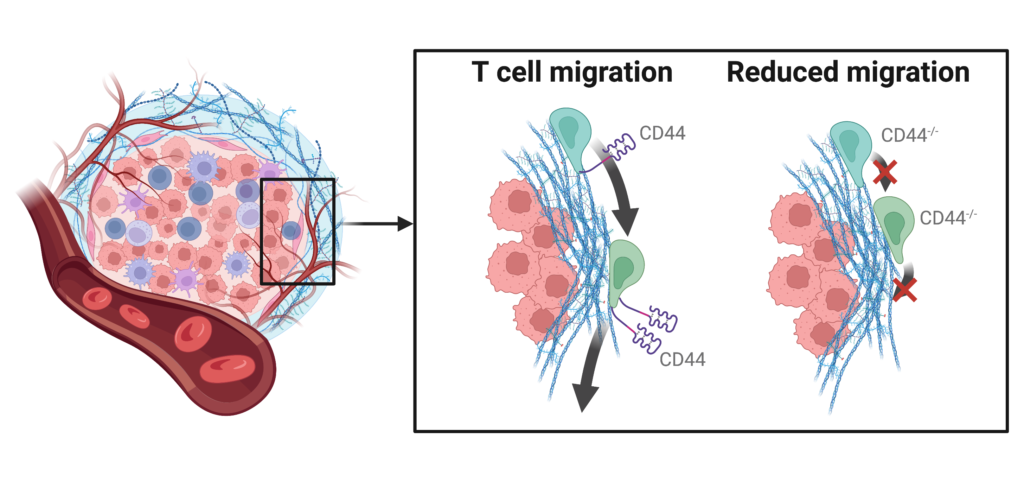
These findings paint a picture of a double edged sword, in which targeting CD44 signaling may shift the balance of myeloid cells toward a more anti-tumorigenic state at the cost of reducing the ability of T cells to navigate through the dense tumor tissue and kill tumor cells. It remains to be determined whether this mechanism explains the failure of the anti-CD44 therapy to treat solid malignancies like glioblastoma. However, even if anti-CD44 did not show any signs of clinical efficacy, targeting other mechanotransducers remains a promising therapeutic approach to improve patient outcomes in glioblastoma and other solid malignancies.